About DDW
Deuterium Depleted Water” (DDW). It is produced using special equipment and undergoes thousands of distillation cycles to reduce the deuterium content in natural water, resulting in a low-deuterium drinking water with specific health benefits.
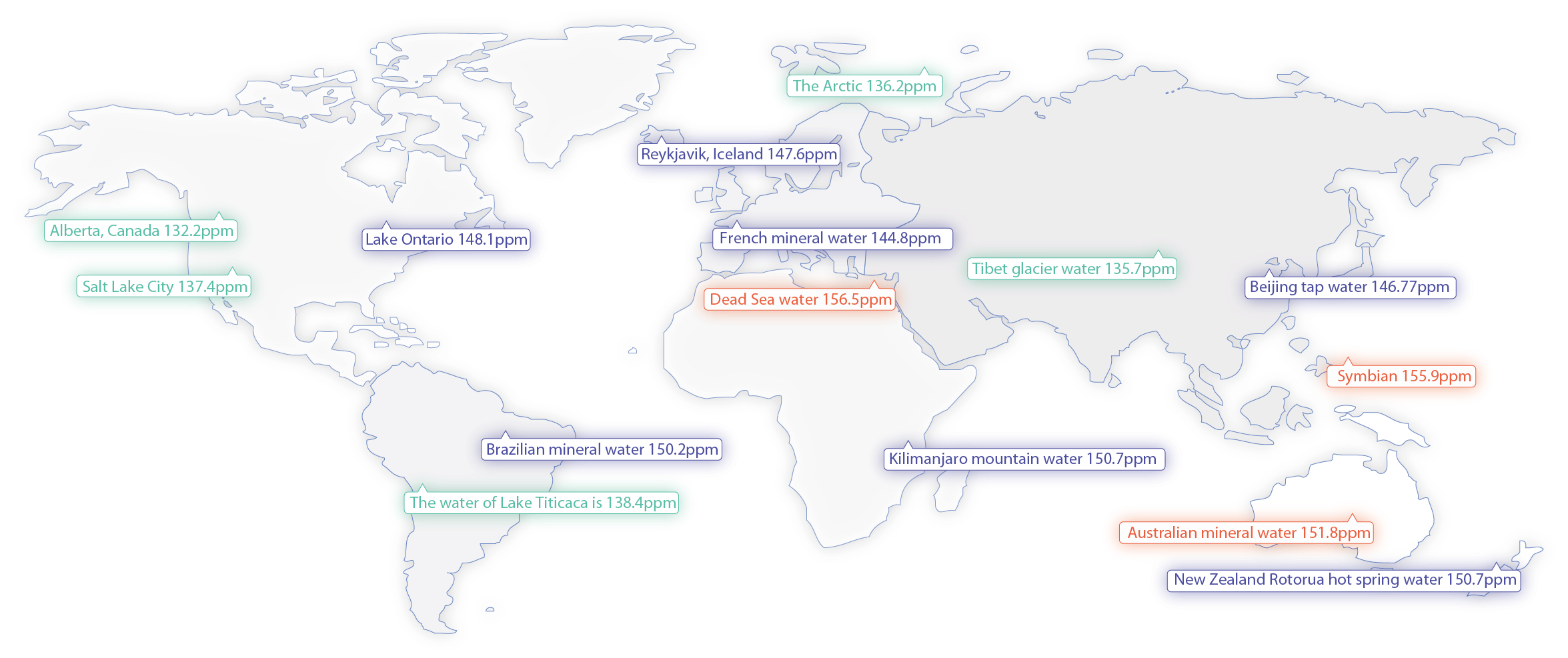
Deuterium is relatively unfamiliar to most people. It is a stable isotope of hydrogen, and hydrogen itself has three isotopes: protium (H), deuterium (D), and tritium (T). While water molecules are typically represented as H2O, in nature, there is no 100% pure “H2O.” The water we commonly consume contains a small amount of deuterium (D), which has one extra neutron compared to hydrogen (H), forming D2O and HDO in the mix. The concentration of deuterium is approximately around 150 PPM (parts per million), meaning about 150 grams of deuterium water in 1 ton of water.

Small Molecular Clusters
In 2013, the Hydration Chemistry Center of the Ukrainian National Academy of Sciences conducted computer simulations on low-deuterium water and found that reducing the deuterium content below that of natural water results in smaller molecular clusters.
Learn moreHigh Surface Tension
The 2013 article titled "Revealing the Secrets of Water Molecules" from the Ukrainian National Academy of Sciences reported that compared to natural water, low-deuterium water exhibits lower viscosity, higher surface tension, reduced density, and enhanced absorbability.
Learn moreLowerChemical Bond Energy
MIT independent researchers have reported that as the hydrogen content within molecules increases (with reduced deuterium), the hydrogen bond energy decreases, significantly enhancing molecular reactivity.
Learn moreThe deuterium content of water at various locations around the world